Levoplant™
Subdermal Contraceptive Implant
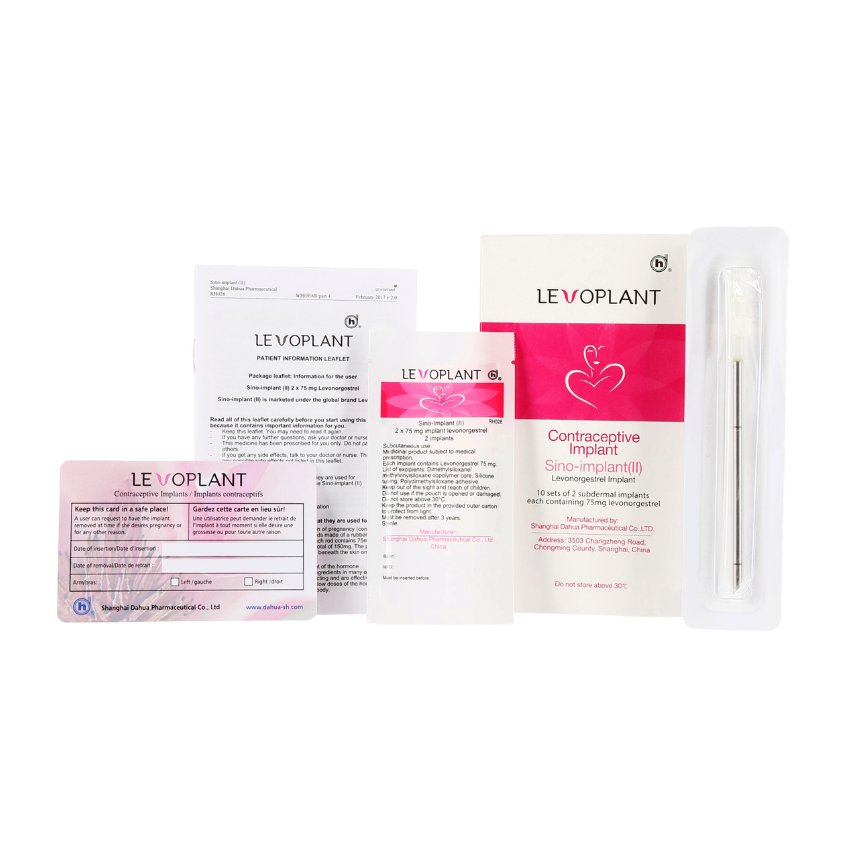
Levoplant™ is a World Health Organization (WHO) Prequalifed subdermal contraceptive implant effective for up to 3 years. Levoplant™ is a highly convenient and acceptable method of contraception.
-
High quality1, affordable, contraceptive implant that is composed of two thin
and flexible silicone rods -
WHO prequalified contraceptive implant2
-
Contains 75 mg of levonorgestrel in each rod (150 mg total)
- More than 99% effective in a 3 year period – less than 1 pregnancy per 100 women over duration of use2 (For heavier women, the effectiveness of Levoplant™ may decrease near the end of the duration. Earlier replacement can be discussed)
- Mechanism of action: Prevents the release of the egg and thickens mucus in the cervical canal to prevent sperm reaching and fertilizing the egg
- Side effects: Irregular bleeding patterns may occur but are not harmful. Abdominal pain, weight changes, breast tenderness, mood changes, migraine can also be experienced.
- Contraindications include unexplained vaginal bleeding, severe liver cirrhosis or liver tumor, history of breast cancer or current deep blood clots
1. Steiner MJ, et al. Randomized trial to evaluate contraceptive efficacy, safety and acceptability of a two-rod contraceptive implant over 4 years in the Dominican Republic. Contracept X. (2019) 1:100006.
2. World Health Organization Department of Sexual and Reproductive Health and Research (WHO/SRH) and Johns Hopkins Bloomberg School of Public Health/ Center for Communication Programs (CCP), Knowledge SUCCESS. Family Planning: A Global Handbook for Providers 4th edition, 2022.
Benefits:
- Estrogen free: avoids estrogen-related side effects that may be experienced with combined hormonal contraceptive methods
- Action to prevent pregnancy is not required once the implant is inserted
- The implant can be removed at any time at the woman’s request and there is no delay in return to fertility
- Almost all women of any age, including those who have just given birth, arebreastfeeding, or women who have had an abortion can use Levoplant™ *if they are medically eligible
Method of Administration:
- Inserted just under the skin of a woman’s upper arm by a trained healthcare provider using a disposable trocar
- Simple insertion and removal takes a few minutes in the clinical setting with local anaesthetic
Available for purchase:
- Levoplant™ – pack of 10
- Levoplant™ – single pack